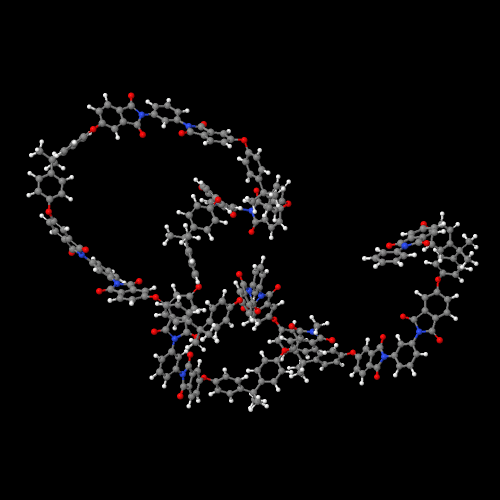
The model above is an image of the pdb model you can view
by clicking here or you can just click on the image itself.
Either way, be sure to close the new window that opens up
with the 3D model in it when you are ready to come back here.
So what's so special about polyimides?
Polyimides are incredibly strong synthetic polymers that are also astoundingly heat and chemical resistant. Their properties are so great that these materials often replace glass and steel in many demanding industrial applications. They may be used for the struts and chassis in some cars as well as some parts under-the-hood because they can withstand the intense heat and corrosive lubricants, fuels, and coolants cars require.
Polyimides are even used in many everyday applications. Who knows, you may even have a few in your house. Do you have polyimides in your kitchen? They are used in many everyday applications such as microwave cookware and food packaging because of their thermal stability, resistance to oils, greases, and fats (YUM!). Even better is their transparency to microwave radiation. They can also be used in circuit boards, insulation, fibers for protective clothing, composites, and adhesives.
By now you have probably guessed that a polyimide is a polymer that contains an imide group.
Yeah, I figured that out, but what is an imide?
I just knew you were going to ask that. An imide is a group in a molecule that has a general structure which looks like this:
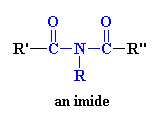
So if the molecular segment shown above were put into a polymer, the product would be, you guessed it, a polyimide. But it's a little more complicated than that. Polyimides can take one of two forms. The first of these structures is a linear structure where the atoms of the imide group are part of a linear chain (see above). The second is a heterocyclic structure where the imide group is part of a cyclic unit in the polymer chain. The cyclic structure is enormously more stable to heat and corrosive environments. This is often the case, that a cyclic form of a functional group is more stable than its linear analog. Care to guess why? Take a look below for schematic structures of the two forms of imides:
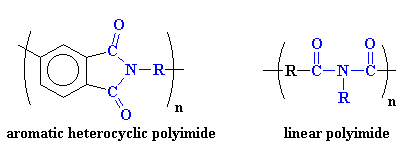
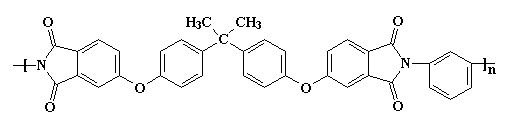
Aromatic heterocyclic polyimides, like the one on the left, are typical of most commercial polyimides, such as Ultem from Sabic (originally invented by G.E. Plastics) and DuPont's Kapton resin. The image at the top of the page is that of Ultem (we believe) and the one below is of Kapton (we think). To examine the 3D version of Kapton, here.
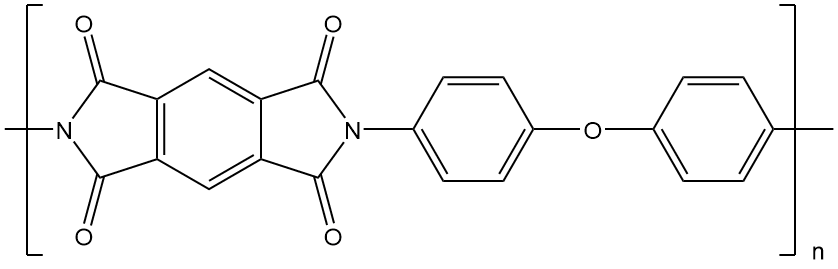
These polymers have such incredible mechanical and thermal properties that they have replaced some metals and glass in high performance applications in the electronics, automotive, and even the aerospace industries. These properties come from strong intermolecular forces between the polymer chains and from the stiffness of the aromatic segments in the repeat units of these polymers.. These forces involve polar interactions, aromatic stacking and most important here, charge transfer complexation.
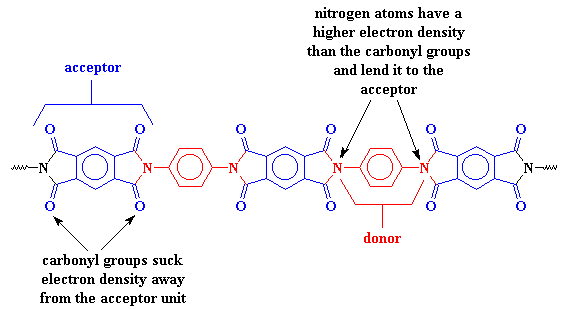
The charge transfer complex works not only between adjacent units in the polymer chain, but also between chains. The chains will stack together like strips of paper, with donors and acceptors paired up and pulling the chains more tightly together. The close stacking is made possible by how flat the aromatic segments are, even those with the imide groups attached. This is good.
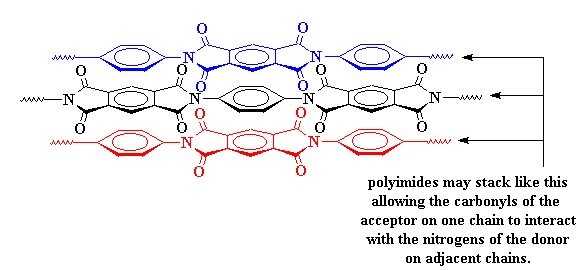
This charge transfer complex holds the chains so tightly that they can't move around very much. When things can't move around on the molecular level, they can't move around in the whole material. This is why polyimides are so strong. It's also why they don't melt or soften except at really high temperatures.
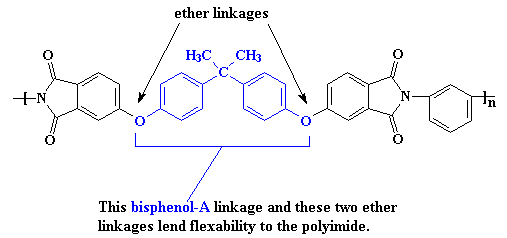
Of course, you can have too much of a good thing. The interactions are so strong, it makes the material difficult to process or do anything with. So it may become necessary to make the polymer a little softer so it can be made into something useful. This is accomplished at the molecular level by using monomers that have more flexible segments. One such is the bisphenol-A derived linkage shown in the polymer segment below. An ether linkage helps with this in the Kapton polyimide.
Another interesting property of polyimides which makes them excellent for use in some construction and transportation industries is how they burn, or rather don't.
Why would you want to build a high-end building
or high-performance car out of something that burns?
Good question, and there's a good answer, too. It is not the burning which industry likes, but the way the burning stops. Polyimides are self extinguishing, which means they may start to burn but then quickly go out. On a molecular level this means that when flame touches the polyimide, a surface char develops which smothers it, blocking it from the oxygen needed for it to burn. Neat, huh?
You might think the high temperature performance and excellent mechanical properties of these polyimides is due to crystallinity. You know that crystalline packing and interactions help with many polymers, and the repeat units of these are pretty symmetrical. You'd be wrong on this one, since most commercial polyimides we're aware of are totally or mostly non-crystalline. Kapton, for example, has no apparent melting transition although it's glass transition temperature (Tg) is somewhere north of 360-400 oC, which is dang high for an organic polymer!
Unfortunately, polyimides are too expensive to build everyday houses and other common objects out of. We use other polymeric materials instead, like wood in home construction. Wood is made of cellulose, of course, which despite already having lots of oxygens in its structure, is more than willing to react with more. Which means that cellulose combines with oxygen from the air to generate heat plus carbon dioxide and water; that is, it burns, as you'd already know if you've ever sat around a campfire on a cool autumn evening. Polyimides might do this, too, if it wasn't for that char layer we just talked about.
Other high performance plastics:| |
And some that aren't plastics: | |
| Polysulfone | Kevlar and Nomex | |
| Polycarbonate | Epoxy Resins | |
| Poly(ether ketone) | Cellulose | |
| Poly(phenylene oxide) | Carbon Fiber | |
| Poly(phenylene sulfide) | Polydicyclopentadiene |

|
Return to Level Two Directory |

|
Return to Macrogalleria Directory |
